We use the Drosophila taste system as a model to study how neural circuits integrate information from our internal and external worlds.
The taste system is a great model to study how the brain integrates different signals to generate flexible behavior. We use our sense of taste to determine what to eat, and our responses to food are profoundly gated by internal signals such as hunger, experience, and reward.
The fruit fly Drosophila offers a wiring diagram of the brain and genetic tools to study neural circuits at single-cell resolution. We combine a broad range of approaches, from molecular and cellular studies to optogenetics, functional imaging, connectomics, behavior, and computational analysis and modeling.
Below are some of our specific research areas.
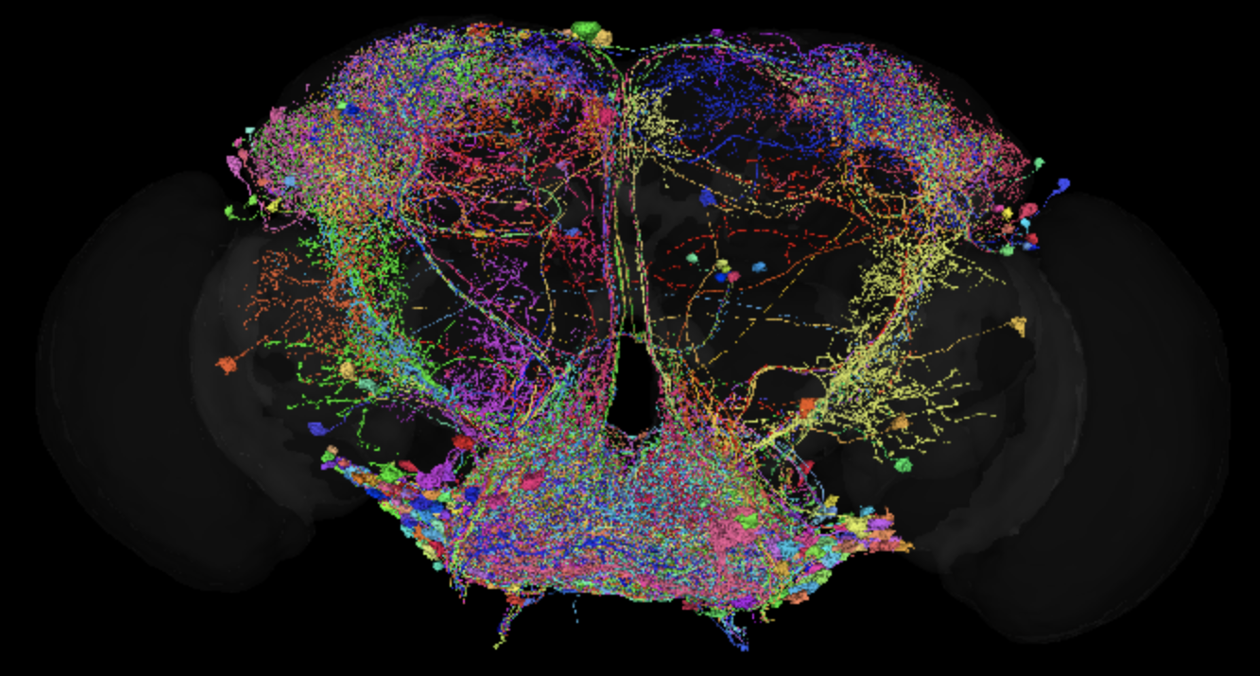
A subset of neurons in the fly brain that process taste information, identified from the connectome (from Walker, Peña-Garcia et al., 2025).
How does the taste circuit transform sensory responses into flexible motor signals?
While taste sensory cells have been well-studied, the downstream circuits that receive and process taste information are largely unknown. We are taking a range of approaches to study how taste circuits transform sensory responses, integrate internal cues, and generate flexible motor signals. We use computational analyses of the synaptic connectome to investigate taste circuit architecture. We use optogenetic manipulations to test how different neurons contribute to behavior, and we use calcium imaging to investigate how they encode and transform sensory information. We also aim to determine where and how modulatory signals such as hunger and experience alter taste processing.
How do animals choose between food options?
Foraging behavior - finding and choosing appropriate food sources in the environment - is critical for survival and provides an ethological model to study the neural mechanisms of decision making. We established a paradigm to study how flies choose between different food patches. We are using quantitative behavioral analyses and modeling to understand how flies explore an environment, learn the value of food options, and use this information to make decisions about what to eat. Our long-term goal is to map these computations onto neural circuits in the brain.
Reward-encoding dopamine neurons in the fly brain that modulate feeding behavior.
How are taste and reward signals integrated in health and disease?
Highly palatable foods like sugar activate reward pathways, and reward perception profoundly modulates food consumption. The dysregulation of reward pathways contributes to eating disorders such as binge eating. As in mammals, food reward in flies is encoded by dopamine neurons, and these reward neurons are also dysregulated in flies that overeat. We are using flies as a model to study how taste, hunger, and reward signals are integrated at the single-cell level, and how reward pathways differentially modulate feeding in health and disease.
Our research is funded by the NIH (NIDCD) and the Whitehall Foundation. We are grateful for their support!